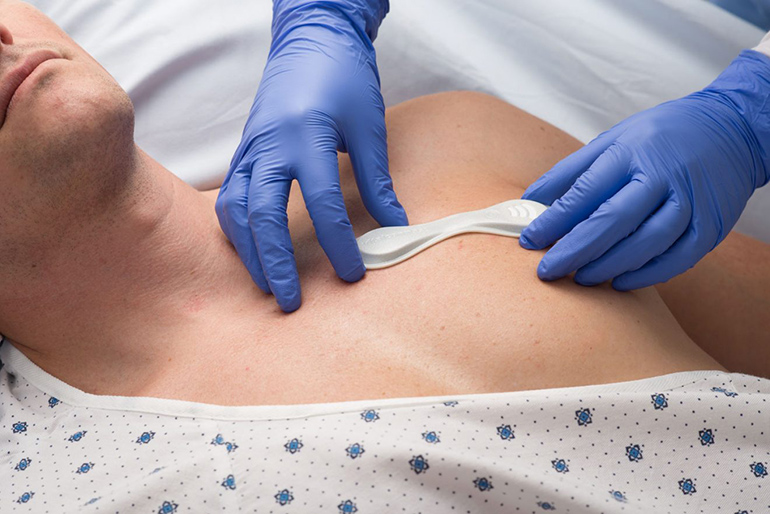
VitalConnect announced that it has received FDA Emergency Use Authorization status for use of its VitalPatch to detect changes in the QT interval of hospitalized patients undergoing drug treatment for COVID-19.
Hydroxychloroquine and chloroquine, used to treat some COVID-19 patients, are associated with risk of prolonged QT interval that can lead to life-threatening arrhythmias. VitalPatch allows clinicians to remotely and continuously monitor patients at risk of QT prolongation due to COVID-19 treatment.
VitalPatch is an FDA-approved device. Besides serving as a single-lead ECG, it monitors seven other physiological parameters continuously, including body temperature, heart rate, heart rate variability, respiratory rate, and blood oxygen saturation levels. It can also integrate with third-party devices to monitor blood pressure, weight, and oxygen saturation. According to the company, it is the first and only biosensor capable of monitoring such a broad set of patient vitals.
VitalPatch can be worn for up to seven days and is powered by a disposable zinc air battery with up to 168 hours of battery life. It measures 120 x 40 mm and is secured to the patient by a hydrocolloid adhesive.
“COVID-19 presents a myriad of symptoms and clinicians need access to medical devices that allow them to monitor and manage those symptoms in real-time in order to create the most appropriate treatment plans for each individual,” said Dr. Joe Roberson, Chief Medical Officer of VitalConnect, in a statement. “The enhancement of the VitalPatch receiving Emergency Use Authorization for QT-interval detection will enable this platform to further support clinicians who are on the frontlines of treating this virus.”