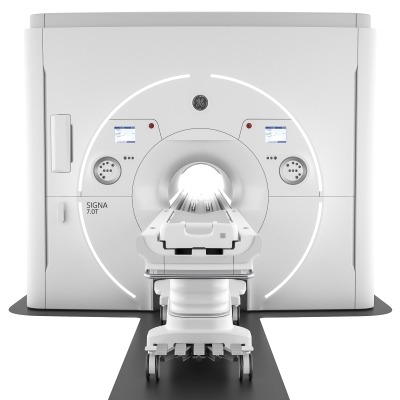
GE Healthcare won FDA clearance for its SIGNA 7.0T MRI scanner, currently the most powerful device available for clinical applications. A high-end scanner’s magnetic field typically tops out at 3 Tesla, but the new device goes all the way up to 7 Tesla to provide an unprecedented look at highly detailed anatomy.
Certainly intended for neurological applications, the SIGNA 7.0T should also be useful for imaging of metabolism, function, and of joints that can’t be properly seen with even a 3 Tesla device. Besides clinical use, the same scanner can just as readily be utilized for research.
In addition to raw power, the SIGNA 7.0T features UltraG gradient technology in the whole-body gradient coil, which helps to get the most out of the powerful magnet. This leads to high resolution, speedier scans, advanced diffusion, and functional MRI (fMRI) imaging of the brain.
SIGNAWorks software works with the hardware to apply deep learning applications to image processing, smart slice positioning, and SilentMR imaging. There are also specialized tools for imaging the knee and cartilage, which may help with close monitoring of the joint during disease progression.
“We are thrilled to add SIGNA 7.0T to our portfolio,” said Jie Xue, president and CEO of GE Healthcare MR, in a press release. “This new scanner is a critical tool in research for neurological disorders like Alzheimer’s disease and mild traumatic brain injury. Now clinicians will have access to the power of ultra-high field imaging combined with the ultra-high-performance gradients to translate research advances into new clinical diagnostic tools and potential treatment options.